Stem Cells
Since the isolation of embryonic stem cells in 1998, labs all over the world have been exploring the possibility of using stem cells to restore damaged or lost tissue. Exciting results are now starting to come in.
What is a stem cell? At the dawn of a human life, a sperm fertilizes an egg to create a single cell destined to become a child. As development commences, that cell begins to divide, producing a small ball of a few dozen cells. At this very early point, each of these cells is identical. We call these cells embryonic stem cells. Each one of them is capable by itself of developing into a healthy individual. In cattle breeding, for example, these cells are frequently separated by the breeder and used to produce multiple clones of valuable offspring.
The exciting promise of these embryonic stem cells is that, because they can develop into any tissue, they may give us the ability to restore damaged heart or spine tissue (figure 24). Experiments have already been tried successfully in mice. Heart muscle cells have been grown from mouse embryonic stem cells and successfully integrated with the heart tissue of a living mouse. This suggests that the damaged heart muscle of heart attack victims might be reparable with stem cells, and that injured spinal cords might be repairable. These very promising experiments are being pursued aggressively. They are, however, quite controversial, as embryonic stem cells are typically isolated from tissue of discarded or aborted embryos, raising serious ethical issues.
Tissue-Specific Stem Cells
New results promise a neat way around the ethical maze presented by stem cells derived from embryos. Go back for a moment to what we were saying about how a human child develops. What happens next to the embryonic stem cells? They start to take different developmental paths. Some become destined to form nerve tissue and, after this decision is taken, cannot ever produce any other kind of cell. They are then called nerve stem cells. Others become specialized to produce blood, still others muscle. Each major tissue is represented by its own kind of tissue-specific stem cell. Now here’s the key point: as development proceeds, these tissue-specific stem cells persist. Even in adults. So why not use these adult cells, rather than embryonic stem cells?
Transplanted Tissue-Specific Stem Cells Cure MS in Mice
In pathfinding 1999 laboratory experiments by Dr. Evan Snyder of Harvard Medical School, tissue-specific stem cells were able to restore lost brain tissue. He and his co-workers injected neural stem cells (immediate descendants of embryonic stem cells able to become any kind of neural cell) into the brains of newborn mice with a disease resembling multiple sclerosis (MS). These mice lacked the cells that maintain the layers of myelin insulation around signal-conducting nerves. The injected stem cells migrated all over the brain, and were able to convert themselves into the missing type of cell. The new cells then proceeded to repair the ravages of the disease by replacing the lost insulation of signal-conducting nerve cells. Many of the treated mice fully recovered. In mice at least, Tissue-specific stem cells offer a treatment for MS.
The approach seems very straight forward, and should apply to humans. Indeed blood stem cells are already routinely used in humans to replenish the bone marrow of cancer patients after marrow-destroying therapy. The
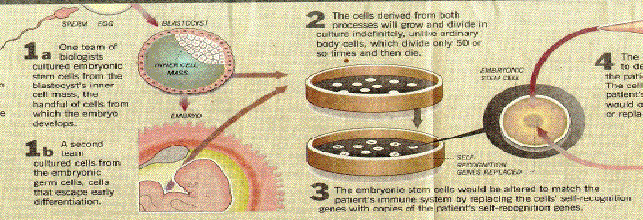 Figure 24a
Figure 24a
Using embryonic stem cells to restore damaged tissue. Embyronic stem cells can develop into any body tissue. Methods for growing the tissue and using it to repair damaged tissue in adults, such as the brain cells of multiple sclerosis patients, heart muscle, and spinal nerves, are being developed.